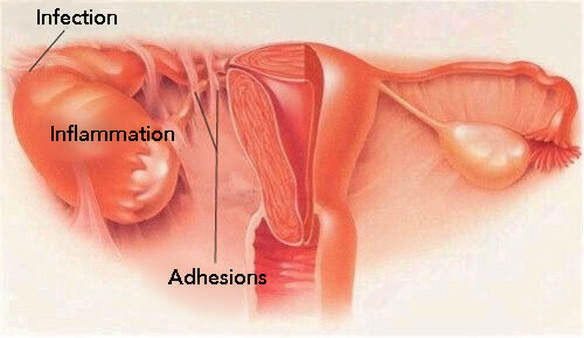
 Diagnosis and management of PID. Pelvic inflammatory disease or PID describes infection and inflammation of the upper genital tract; this may involve the endometrium, fallopian tubes and/or ovaries, as well as the surrounding peritoneum. PID is a common cause of morbidity and accounts for one in 60 general practitioner consultations by women under the age of 45 years.1 With a single episode of PID, the incidence of subfertility is 20% due to tubal and ovarian adhesions. There is a risk of tubo-ovarian abscesses, chronic pelvic pain and a significantly increased risk of ectopic pregnancy.1 Repeated episodes of PID may increase the risk of permanent tubal damage exponentially. Clinically more severe disease is associated with a greater risk of sequelae.2 Aetiology Most cases of PID result from a vaginal or cervical sexually transmitted infection; this may be asymptomatic. Subsequently there is direct ascent of micro-organisms from the vagina or cervix to the upper genital tract.2 On occasion PID may lead to Fitz-Hugh-Curtis syndrome, which describes the situation where infection spreads along the upper peritoneum to the liver capsule causing perihepatic ‘violin string’ adhesions. It is thought that Chlamydia trachomatis and Neisseria gonorrhoeae are responsible for most cases of PID.3 Other potential micro-organisms include Gardnerella vaginalis, Mycoplasma hominis and Trichomonas vaginalis.1 Human immunodeficiency virus (HIV) infection has been found to be associated with an increased incidence of Chlamydia trachomatis infection. Women with HIV infection have an increased risk of progression to PID and tubo-ovarian abscess formation.1 Sexually active women that are in their teens or early twenties are at increased risk of PID, as are women from lower socio-economic backgrounds. Presence of a sexually transmitted infection, previous pelvic infections, unprotected sexual intercourse, sexual intercourse at an early age and multiple sexual partners are risk factors for PID.4 Less commonly instrumentation of the cervix and/or of the uterus can result in endogenous bacteria inoculating the endometrium and consequently leading to PID. The relative risk of PID is higher in intra-uterine device users however the absolute risk remains very low, in the order of one in one thousand.2 Clinical features and diagnosis At presentation, women with PID may range from asymptomatic to being systemically unwell. Ascending infection from the endocervix may cause endometritis, salpingitis, parametritis, oophoritis, tubo-ovarian abscess and/or pelvic peritonitis.1 PID attributed to chlamydial infection tends to be asymptomatic and diagnosis is retrospective when a patient presents with subfertility due to tubal adhesions. In contrast, PID caused by Neisseria gonorrhoeae tends to be acute.1 The most common presenting complaint is acute lower abdominal or pelvic pain in association with fever in a sexually active woman.2 Bilateral lower abdominal pain is typical with deep dyspareunia; abnormal vaginal or cervical discharge which is often purulent and abnormal vaginal bleeding, including post coital or intermenstrual bleeding and menorrhagia are other associated features.5 Nausea and vomiting may be a feature in PID. On examination, there may be a mild tachycardia and fever. There is likely to be bilateral lower abdominal tenderness; rebound tenderness and guarding may be elicited. If there is right upper quadrant tenderness Fitz-Hugh-Curtis syndrome should be suspected. Bimanual examination is likely to reveal adnexal tenderness and cervical excitation; there may be a tubo-ovarian abscess that may be appreciated on bimanual palpation.2 Mucopurulent discharge from the cervix or vagina may be seen on speculum examination, and high vaginal and endocervical swabs should be taken. An elevated white cell count and C-reactive protein supports the diagnosis of PID in conjunction with the clinical findings.5 Liver function tests may be deranged in the presence of Fitz-Hugh-Curtis syndrome and in acute cases. Differential diagnoses may include appendicitis, diverticulitis, urinary tract infection, ectopic pregnancy, ovarian torsion or rupture. A pregnancy test is required in all women of childbearing age. A diagnosis of PID should be made on clinical grounds. Moreover negative swab results do not rule out a diagnosis of PID.2,5 A delay in diagnosis or treatment of PID can result in long-term sequelae, such as chronic pelvic pain and tubal infertility. If a diagnosis of PID is made, testing for other sexually transmitted infections is recommended.6 Hospital admission is recommended if there is a suspicion of ectopic pregnancy or a surgical emergency such as acute appendicitis, a tubo-ovarian abscess is suspected or there is diagnostic uncertainty. Severity of symptoms and signs such as nausea, vomiting and a fever greater than 38°C may indicate that hospital admission may be necessary.3 Transvaginal ultrasound examination is not helpful in diagnosis but may demonstrate tubo-ovarian abscesses. If classic findings of PID are noted on ultrasound, no further imaging is required.7 If additional imaging is warranted MRI is recommended over CT because its overall accuracy is greater than 93% and does not carry the additional risk of ionising radiation.7 Management Since there are no widely accepted clinical criteria for PID, early empirical treatment is common. Antibiotic regimens for the treatment of PID must cover Chlamydia trachomatis, Neisseria gonorrhoeae, anaerobes, Gram-negative aerobes and streptococci.1 This provides cover for the most likely sexually transmitted infections as well as endogenous vaginal and lower gastrointestinal flora. Preferably treatment should be started as early as possible to minimise the risk of future ectopic pregnancy and tubal infertility. However, seeking medical attention may be delayed, as clinical features may not be apparent or appreciated by patients in the subclinical stage. Those women with HIV may present with more severe symptoms of PID, it may be appropriate to manage these patients on an outpatient basis however parenteral treatment may be needed. Outpatient therapy is as effective as inpatient treatment for patients with clinically mild to moderate PID, however hospitalisation should be considered in selected cases.6,7 Antibiotic regimens that may be considered for outpatients are as follows: ofloxacin 400 mg orally twice daily plus oral metronidazole 400 mg twice daily, both for 14 days OR ceftriaxone 500 mg as a single intramuscular dose, plus oral doxycycline 100 mg twice daily and oral metronidazole 400 mg twice daily, both for 14 days. Ofloxacin should be avoided in patients who are at high risk of gonococcal PID because of increasing quinolone resistance in the UK.1 A clinical review at 72 hours is recommended in those women with a moderate or severe clinical presentation of PID. There should be demonstrable clinical improvement at this review and if there has been little improvement, hospital admission or an alternative diagnosis should be considered.5 At this review antibiotic sensitivities from swab results are likely to be available. Treatment should be continued even in the absence of negative swab results. Ideally sexual partners within the last six months should be seen in a genito-urinary medicine clinic so that screening, treatment and contact tracing may be implemented.5 Hospital admission should be considered if there are severe symptoms and signs of PID, if oral therapy does not lead to resolution of symptoms, if there is concern about the presence of a tubovarian abscess or if there is diagnostic uncertainty.2 Surgical management includes laparoscopy which may help early resolution of the disease by dividing adhesions and draining pelvic abscesses. It has been suggested that ultrasound- guided aspiration of pelvic fluid collections may be equally effective. In the case of Fitz-Hugh-Curtis syndrome adhesiolysis may be performed, however there is no evidence as to whether this is superior to antibiotic therapy.5 In a woman presenting with PID it may be necessary to consider removing any contraceptive intrauterine device after appropriate counselling about the risk of pregnancy. In this situation emergency hormonal contraception may be appropriate.1 Prognosis PID may be asymptomatic but may cause substantial morbidity. A low threshold for empirical treatment of PID is recommended because of the lack of definitive clinical diagnostic criteria and because the potential consequences of not treating of PID.4 Patients should be advised to avoid unprotected intercourse until treatment and follow-up including contact tracing has been completed.1 Clinical review at 72 hours is recommended in those women with a moderate or severe clinical presentation to ensure improvement. Further review at 2-4 weeks may be helpful to ensure clinical response in addition to screening and treatment of sexual contacts.5 Preventing gonococcal and chlamydial infections represents the most effective means of reducing the incidence of PID. Interestingly over the past decade the incidence of pelvic infection in England has fallen by over a half.3 Educating women about the sequelae of untreated sexually transmitted infections as well as emphasising how to prevent transmission of such infections is important. For example, The National Chlamydia Screening Programme (NCSP) is targeted at the highest risk group for chlamydia infection in England who are sexually active. This group may be offered opportunistic screening in general practice.8 Furthermore, women on hormonal contraception presenting with breakthrough bleeding should be screened for sexually transmitted infections.
0 Comments
Leave a Reply. |
Dr. Jaspal SachdevDr.Jaspal Singh Sachdev is the Resident Consultant and Head of Unit of Obstetrics and Gynaecology at Park City Medical Centre a Ramsay Sime Darby Hospital. Archives
December 2016
Categories |

 RSS Feed
RSS Feed
